Tag: Gene Therapy
First Gene Therapy for Children With Metachromatic Leukodystrophy Approved by FDA
Approval is for a single-dose gene therapy infusion
Gene Therapy for Sickle Cell Disease Likely Cost-Effective at <$2 Million
Incremental cost-effectiveness ratios were $126,000 and $281,000 per QALY for two simulation models from a societal perspective
First Gene-Editing Therapies for Sickle Cell Disease Approved by FDA
In both therapies, stem cells are removed from a patient's blood for treatment
FDA Advisors Say New Gene Therapy for Sickle Cell Disease Is Safe
Decision clears the way for full approval of the treatment by early December
FDA Advisers to Weigh New Gene Therapies for Sickle Cell Anemia
Advisers will consider whether more research is needed into potential unintended consequences of the new therapies
Racial, Ethnic Differences ID’d for Idecabtagene Vicleucel in Multiple Myeloma
Non-Hispanic Black patients more likely to develop cytokine release syndrome; best overall response rate was lower in Hispanics
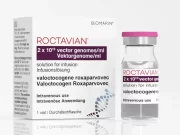
FDA Approves First Gene Therapy for Severe Hemophilia A
Roctavian is a viral vector that carries the gene for factor VIII
FDA Approves First Gene Therapy to Treat Duchenne Muscular Dystrophy
The recombinant gene therapy is administered in a single intravenous dose
Gene Therapy Not Cost-Effective for Sickle Cell by Conventional Measures
The inequality aversion parameter would need to be 0.90 for gene therapy to be preferred per distributional cost-effectiveness analysis standards
Benefits of Valoctocogene Roxaparvovec Persist in Hemophilia A
The mean annualized treated bleeding rate decreased by 84.5 percent from baseline to 104 weeks